Global Sterile Injectables Market Growth to Reach $1.07B by 2033
Rising demand for safe, efficient injectable therapies and advanced drug delivery is driving growth and innovation in the sterile injectables sector worldwide.
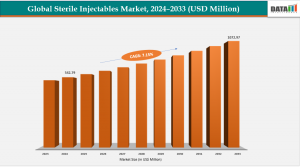
The market shows steady growth at 7.15% CAGR, reflecting strong adoption of sterile injectables, technological advances, and strategic expansions by pharma players globally.”
AUSTIN, TX, UNITED STATES, February 26, 2026 /EINPresswire.com/ -- The Global Sterile Injectables Market reached US$ 542.79 Million with a rise of US$ 578.61 Million in 2024 and is expected to reach US$ 1072.97 Million by 2033, growing at a CAGR of 7.15% during the forecast period 2025-2033.— DataM Intelligence
The market is expanding in the US due to major investments like Jubilant HollisterStier’s $300M sterile injectable capacity expansion and strong FDA approvals for complex injectables, while in Japan, strategic partnerships such as Otsuka–Towa’s collaboration and increased hospital demand for injectable therapies are driving growth.
Download your exclusive sample report today: (corporate email gets priority access):https://www.datamintelligence.com/download-sample/sterile-injectable-market
Key Industry Development-
United States: Key Industry Developments
→ January 2025: Akums Drugs and Pharmaceuticals launched a new sterile facility in the U.S. market for producing lyophilised injectables, vials, ampoules, and eye/ear drops, enhancing capacity for high-demand sterile products amid rising chronic disease treatments.
→April 2025: Argenx received FDA approval for VYVGART Hytrulo prefilled syringe, a sterile injectable for autoimmune conditions, boosting the CMO market through advanced fill-finish technologies and self-administration convenience.
Japan: Key Industry Developments
→September 2025: Takeda Pharmaceuticals expanded sterile injectables production in Japan with new biologics lines, focusing on oncology and diabetes therapies to meet Asia Pacific's 7.5% CAGR growth in the sector.
Strategic Mergers and Acquisitions:
PAI Pharma acquired Nivagen Pharmaceuticals (January 2026): PAI Pharma, backed by Olympus Partners, bought the California-based firm specializing in ready-to-use sterile injectables. Value undisclosed. Strategic rationale: Expand domestic manufacturing, bolster supply chain resilience, and tackle US hospital drug shortages.
Thermo Fisher Scientific acquired Sanofi’s Ridgefield site (announced July 2025, completed September 2025): Thermo Fisher purchased Sanofi’s sterile fill-finish facility in New Jersey (terms undisclosed). The site continues producing for Sanofi while serving other pharma/biotech clients. Strategic rationale: Boost US manufacturing capacity amid rising demand.
Major Players:-
Pfizer Inc. | F. Hoffmann-La Roche Ltd | Novartis Pharmaceuticals Corporation | Johnson & Johnson | Sanofi | AstraZeneca | Amgen Inc. | Bristol-Myers Squibb Company | GSK | Takeda Pharmaceuticals U.S.A., Inc.
Pfizer Inc. – Pfizer holds 14% of the sterile injectables market, driven by a broad portfolio of vaccines, oncology biologics, and acute‑care injectables. Its acquisition of Hospira expanded sterile manufacturing capacity, reinforcing leadership in hospital injectables and biosimilars across global markets.
Johnson & Johnson – J&J captures 11% share through its strong immunotherapy, biologics, and high‑potency sterile products. Its global reach and robust R&D in biologics and immunology give competitive advantage in injectable therapies critical to hospital and specialty care.
Novartis Pharmaceuticals Corporation – Novartis, via its Sandoz division, commands significant market presence with biosimilars and complex sterile injectables. Its focus on affordable biologics, regulatory compliance, and advanced manufacturing strengthens access in major regions.
Sanofi – Sanofi maintains a notable share with its sterile vaccine and specialty care injectables. Strategic CDMO partnerships and expansion of oncology and immunology pipelines enhance production capabilities and market penetration, especially in vaccines and rare disease therapies.
AstraZeneca – AstraZeneca’s sterile injectable competitive edge stems from its oncology and biologics portfolio approved in numerous countries. The company’s investment in advanced formulations and global commercial strategy ensures strong hospital and therapeutic adoption.
"Secure your 30% discount - get this report before the offer expires."
:https://www.datamintelligence.com/buy-now-page?report=sterile-injectable-market (Purchase 2 or more Repots and get 50% Discount)
Market Segmenatation-
Molecule Type: In the United States sterile injectables market, large molecules such as biologics and monoclonal antibodies dominate the revenue share because of advanced biopharmaceutical development and high adoption for oncology and chronic diseases, reflecting the preference for targeted therapies over conventional small molecules. In Japan, large molecule sterile injectables also hold the largest segment, particularly within complex biologics and high‑value formulations, supported by strong R&D and demand in hospital settings, while small molecules continue to serve established therapeutic uses.
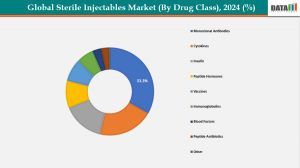
Drug/Drug Class: For the US market, monoclonal antibodies form the largest drug class segment within sterile injectables, driven by extensive use in cancer and autoimmune treatments and reflecting a high share of development pipelines. In Japan, segmentation by drug class shows a similar trend with oncology injectables (including biologics) leading demand alongside other therapeutic classes like anti‑infectives and cardiovascular injectables, tailoring to the country’s aging population and hospital treatment patterns.
Therapeutic Application: In the US sterile injectables market, oncology remains the top therapeutic segment due to high cancer prevalence and the need for IV‑administered therapies, followed by other indications such as metabolic and infectious diseases. Japan’s therapeutic segmentation reflects significant hospital‑based use for conditions like cancer and chronic disease management, with demand patterns shaped by demographic trends and clinical practice, where hospital administrations drive most sterile injectable usage.
Route of Administration: Intravenous (IV) continues to be the most significant route in the US as hospitals and specialty clinics prefer it for rapid systemic delivery in critical care and oncology settings, with subcutaneous (SC) and intramuscular (IM) formats growing for homecare and chronic therapies. In Japan, the IV route also dominates medical practice for sterile injectables in acute and inpatient care, while SC and IM routes support broader applications in outpatient and chronic therapies as healthcare delivery models evolve.
Market Drivers:-
• High Prevalence of Chronic & Complex Diseases: The rising number of chronic conditions such as cancer, diabetes, autoimmune disorders, and cardiovascular diseases fuels demand for sterile injectable therapies that deliver rapid, targeted treatment, especially in hospital and critical‑care settings.
• Strong Biologics and Biosimilars Expansion: Rapid adoption of biologic drugs (including monoclonal antibodies and complex peptide therapeutics) and increasing FDA approvals boost the sterile injectables segment, as many of these therapies require sterile parenteral delivery.
• Robust R&D and Manufacturing Infrastructure: The U.S. leads globally in sterile injectable production capacity and pharmaceutical innovation, supported by a well‑established pharmaceutical ecosystem, high R&D spending, and transparent regulatory frameworks that facilitate complex drug development and commercialization.
• Escalating Hospital & Emergency Care Utilization: Over 60 % of hospital‑administered medications in the U.S. are sterile injectables driven by preference for injectable therapies in emergency settings, oncology, immunology, and acute care.
• Outsourcing and CDMO Growth: Increasing outsourcing of sterile fill‑finish services to Contract Development and Manufacturing Organizations (CDMOs) accelerates market growth and supports faster time‑to‑market for complex injectable drugs.
Regional insights:-
North America: 34.9% (Largest share globally primarily driven by the United States with robust healthcare infrastructure and high biologics & specialty injectable uptake).
Asia Pacific: 28% (Rapid growth, supported by expansion in China, India, and increasing injectable demand; Japan contributes as one of the major APAC sub‑markets).
• Japan specific: Japan accounted for 7.2% of the global sterile injectables CDMO market in 2024 and continues to grow steadily due to an aging population and advanced healthcare system.
Europe: 26% (Steady market with strong biosimilar adoption and expansive chronic/oncology care injection usage).
Middle East & Africa: 8% (Emerging market with rising healthcare investment and injectable demand).
Latin America: 3.5% (Smaller share but growing due to expanding hospital access).
Get Customization in the report as per your requirements:https://www.datamintelligence.com/customize/sterile-injectable-market?prtk
Conclusion :
Rising healthcare needs, technological advances, and strategic collaborations are set to drive robust growth in the sterile injectables market, making it a key focus for pharmaceutical and healthcare stakeholders.
Related Reports:-
1. Electrosurgical Pencils Market
2. Patient Return Electrodes Market
Sai Kiran
DataM Intelligence 4market Research LLP
+1 877-441-4866
email us here
Visit us on social media:
LinkedIn
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.